
Catalog # WP1731
WAVE1 (N-terminal region) Antibody
Rabbit Polyclonal
| ELISA | 1:2000 |
| ICC | 1:200 |
| WB | 1:1000 |
Size 100 μl
Species Reactivity Hu, Rt, Ms
MW 80 kDa
The Wiskott–Aldrich syndrome protein (WASP) family is involved in various pathways that regulate actin cytoskeletal organization. This family includes WASP, N-WASP, and three WAVE/SCAR isoforms, WAVE1, 2, and 3. WAVE proteins play key roles in actin-mediated cell events, such as membrane ruffling and lamellipodia formation. WAVEs contain an N-terminal WAVE homology domain, a basic domain, a Proline-rich region, and carboxy terminal verprolin, cofilin, and acidic (VCA) region. WAVEs are thought to act downstream of the Rac GTPase, connecting Rac activation to induction of Arp 2/3-mediated actin polymerization. Regulation of WAVE activity can occur through tyrosine phosphorylation. Src phosphorylation of WAVE1 at Tyr-125 enhances binding to the Arp2/3 complex, and is required for WAVE inhibition of Arp2/3-mediated stress fiber formation. By contrast, WAVE2 phosphorylation of Tyr-150 by Abl may enhance Arp2/3 complex actin nucleation and microspike formation in fibroblasts. Thus, site-specific tyrosine phosphorylation may be important for controlling specific activities of WAVE proteins.
References
Ardern, H. et al. (2006) Cell Motil. Cytosk. 63:6.
Leng, Y. et al. (2005) PNAS 102(4):1098.
Miki, H. et al. (1999) J Biol. Chem. 274(39):27605.
Suetsugu, S. et al. (1999) Bioch. Biophs. Res. Comm. 260:296.
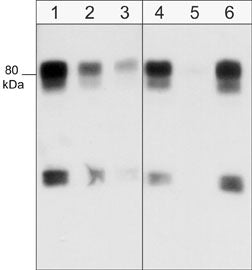
Western blot of human SYF cSrc-transformed cells. Blots were were probed with anti-WAVE1 (N-terminal region) at a dilution of 1:1000 (lane 1), 1:2000 (lane 2) or 1:4000 (lane 3). In addition, the antibody was used in the absence (lane 4) or presence of blocking peptides, WAVE1 (N-terminal region) peptide (lane 5) or WAVE2 (Central region) peptide (lane 6).

Immunocytochemical labeling of phosphorylated WAVE in pervanadate-treated mouse C2C12. The cells were labeled with rabbit polyclonal WAVE1 (N-terminal region) and WAVE (Tyr-125) antibodies, then the antibodies were detected using appropriate secondary antibodies conjugated to Cy3.
*For more information, see UniProt Accession Q92558
The products are are safely shipped at ambient temperature for both domestic and international shipments. Each product is guaranteed to match the specifications as indicated on the corresponding technical data sheet. Please store at -20C upon arrival for long term storage.
*All molecular weights (MW) are confirmed by comparison to Bio-Rad Rainbow Markers and to western blot mobilities of known proteins with similar MW.
This kit contains:








































