
Catalog # IP1861
IκBα (C-terminus) Antibody
Rabbit Polyclonal
| ELISA | 1:2000 |
| WB | 1:1000 |
Size 100 μl
Species Reactivity Hu, Rt, Ms
MW 38 kDa
The NF-κB/Rel transcription factors are present in the cytosol in an inactive state complexed with the inhibitory IκB proteins. Activation of IκBα occurs through both serine and tyrosine phosphorylation events. Activation through phosphorylation at Ser-32 and Ser-36 is followed by proteasome-mediated degradation, resulting in the release and nuclear translocation of active NF-κB. This pathway of IκBα regulation occurs in response to various NF-κB-activating agents, such as TNFα, interleukins, LPS, and irradiation. An alternative pathway for IκBα regulation occurs through tyrosine phosphorylation of Tyr-42 and Tyr-305. Tyr-42 is phosphorylated in response to oxidative stress and growth factors. This phosphorylation can lead to degradation of IκBα and NF-κB-activation. In contrast, Tyr-305 phosphorylation by c-Abl has been implicated in IκBα nuclear translocation and inhibition of NF-κB-activation. Thus, tyrosine phosphorylation of IκBα may be an important regulatory mechanism in NF-κB signaling.
References
Bui, N.T. et al. (2001) J Cell Biol 152(4):753.
Finco, T.S. et al. (1994) Proc. Natl. Acad. Sci. USA 91:11884.
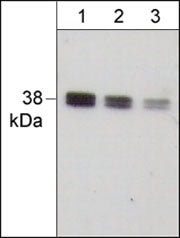
Western blot image of human A431. The Blots were probed with anti-IκBα (C-term.) polyclonal antibody at a dilution of 1:500 (lane 1), 1:1000 (lane 2), and 1:2000 (lane 3).
*For more information, see UniProt Accession P25963
The products are are safely shipped at ambient temperature for both domestic and international shipments. Each product is guaranteed to match the specifications as indicated on the corresponding technical data sheet. Please store at -20C upon arrival for long term storage.
*All molecular weights (MW) are confirmed by comparison to Bio-Rad Rainbow Markers and to western blot mobilities of known proteins with similar MW.
This kit contains:








































