
Catalog # CP1151
Cofilin 1 (Ser-3), phospho-specific Antibody
Rabbit Polyclonal
| ELISA | 1:2000 |
| ICC | 1:50 |
| WB | 1:1000 |
Size 100 μl
Species Reactivity Hu, Rt, Ms, Ck
MW 19 kDa
Members of the ADF/cofilin (AC) family are actin-severing proteins that regulate actin remodeling during cellular events such as cell migration, cytokinesis, phagocytosis, endocytosis, axon development, and immune cell activation. In mammals, there are three members of the AC family, muscle-specific cofilin (Cofilin 2), non-muscle cofilin (Cofilin 1), and ADF. In humans, cofilin 1 and ADF have 72% identity, with the major amino acid differences found in the C-terminal region. Regulation of cofilin activity can occur through serine phosphorylation. Activation of cofilin kinases, LIMK1 or LIMK2, leads to phosphorylation of cofilin at serine 3. This phosphorylation disrupts cofilin binding to actin in vitro and in vivo. Multiple phosphatases, PP1, PP2A, PP2B, slingshot, and chronophin can dephosphorylate Ser-3 and activate actin binding. Thus, Ser-3 phosphorylation is a major site for the regulation of cofilin activity.
References
Maciver, S.K. & Hussey, P.J. (2002). Genome Biol. 3(5):3007.
Aizawa, H., et al. (2001). Nature Neurosci. 4(4):367.
Bamburg, J.R. (1999). Annu Rev Cell Dev Biol. 15:185.
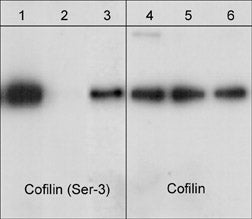
Western blot of cofilin in jurkat cells. The blots were untreated (lanes 1 & 4) or treated (lanes 2, 3, 5 & 6) with lambda phosphatase. In lanes 3 & 6, the phosphatase was inhibited with phospho-Cofilin 1 (Ser-3) peptide. The blots were probed with anti-Cofilin 1 (Ser-3) phospho-specific (lanes 1-3) or anti-Cofilin 1 (N-terminus) (lanes 4-6).

Immunocytochemical labeling in chick dorsal root ganglion neurons using anti-Cofilin (N-terminus; CP1131), anti-Cofilin (Ser-3; CP1151), anti-βIII-Tubulin (C-terminus; TP1691) and anti-β-Tubulin (TM1541) antibodies. (Images provided by Dr. Diane Snow, Department of Anatomy & Neurobiology, University of Kentucky).
*For more information, see UniProt Accession P23528
The products are are safely shipped at ambient temperature for both domestic and international shipments. Each product is guaranteed to match the specifications as indicated on the corresponding technical data sheet. Please store at -20C upon arrival for long term storage.
*All molecular weights (MW) are confirmed by comparison to Bio-Rad Rainbow Markers and to western blot mobilities of known proteins with similar MW.
Product References:
Rufanova, V.A. et al. (2009) Cell Health and Cytoskel. 1:17. (WB: Glomerular mesangial cell)Giacobbe, A. et al. (2016) Oncogene. 35(12):1602-8. (WB: MCF-7)
Amelio, I. et al. (2012) J Cell Biol. 199(2):347. (WB: human primary epidermal keratinocytes)
Couch, B.A. et al. (2010) J Alzheimers Disease. 20:1003. (WB: mouse hippocampus, fasudil)
This kit contains:








































