
Catalog # EP1911
EGFR (Ser-967), phospho-specific Antibody
Rabbit Polyclonal
| ELISA | 1:2000 |
| ICC | 1:200 |
| WB | 1:1000 |
Size 100 μl
Species Reactivity Hu, Rt, Ms
MW 180 kDa
The epidermal growth factor receptor (EGFR) is a transmembrane glycoprotein with an extracellular ligand-binding domain and a cytoplasmic domain with intrinsic tyrosine kinase activity. The cytoplasmic domain has a C-terminal region with multiple autophosphorylation sites (Tyr-992, 1068, 1086, 1148, and 1173). These sites are important for downstream signaling and rapid internalization. In addition, EGFR activation leads to c-Src mediated phosphorylation of Tyr-845 and Tyr-1101. The former site is required for mitogenic responses to EGFR activation, while the latter may be an SH2 binding site. Phosphorylation of EGFR on serine and threonine residues is thought to represent a mechanism for regulation of receptor kinase activity and internalization. These sites include a PKC site (Thr-654), CAMKII sites (Ser-1046, 1047, 1057, and 1142), and constitutively phosphorylated sites (Ser-967 and Ser-1002). Thus, the regulation of EGFR activity involves a complex series of phosphorylation events at multiple sites throughout the intracellular portion of the receptor.
References
Boeri Erba, E. et al. (2005) Mol. Cell. Prot. 4:1107.
Carpenter, G. (2000) Bioessays 22:697.
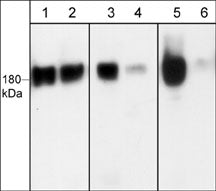
Western blot image of human A431 cells treated with Calyculin A (100 nM) for 30 min. Blot lanes were untreated (lanes 1, 3, & 5) or treated with lambda phosphatase (lanes 2, 4, & 6) then were probed with anti-EGFR (a.a. 961-972) (lanes 1 & 2), anti-EGFR (Ser-967) (lanes 3 & 4), and anti-EGFR (Ser-1142) (lanes 5 & 6).

Immunocytochemical labeling in A431 cells untreated or treated with Calyculin A (50 nM) for 15 min. Cells were labeled with anti-EGFR (a.a. 961-972) or anti-EGFR (Ser-967) antibodies. The specificity of labeling was demonstrated for each antibody by blocking the signals with their respective blocking peptides, EGFR (a.a. 961-972) (EX1875) and phospho-EGFR (Ser-967) (EX1915).
*For more information, see UniProt Accession P00533
The products are are safely shipped at ambient temperature for both domestic and international shipments. Each product is guaranteed to match the specifications as indicated on the corresponding technical data sheet. Please store at -20C upon arrival for long term storage.
*All molecular weights (MW) are confirmed by comparison to Bio-Rad Rainbow Markers and to western blot mobilities of known proteins with similar MW.
This kit contains:




